Li-ions can be intercalated in the layers, which each are approx. One of the most commonly used anode material is graphite. There are three main categories for anode types which are, insertion like graphite, conversion, and lithium alloys like Li x M y (M = Al, Ga, In, Si, Ge, Sn, Pb, Sb, and Bi). Carbonate-based electrolytes have the advantage, that they dissolve Li-salts and they can also form protective layers on graphitic anode materials. In most commercially used batteries lithium salts (LiPF 6) are dissolved in a non aqueous organic solvent like for example dimethyl carbonate (DMC) or ethylene carbonate (EC). High ionic conductivity (10 −3 S cm −1), low electronic conductivity (10 −10 S cm −1), thermal stability, cheap production, and environmental friendliness are the main requirements for a good electrolyte. Electrolytes play a major role in the safety of LIBs. 
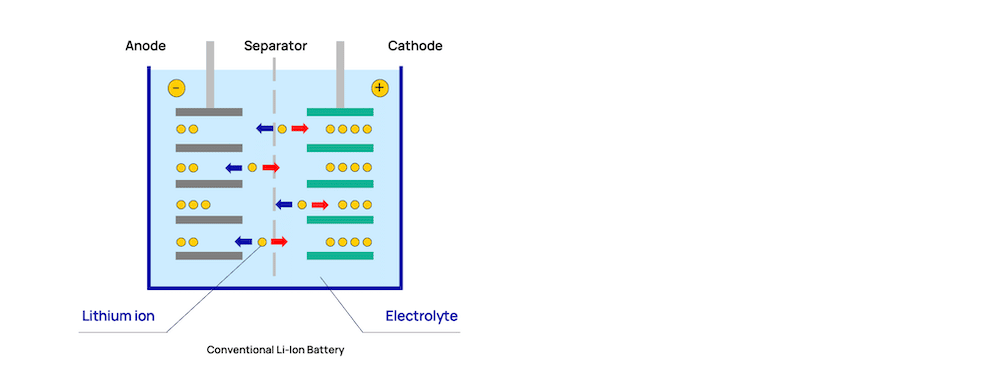
The electrolyte in a battery is the media through which the lithium ions move back and forth between the oppositely charged electrodes. In Li-NMC cobalt has the oxidation state Co 3+ and is replaced gradually with Ni 2+ and Mn 4+.The most commonly used stoichiometry of LiNi xMn 圜o zO 2 (x,y,z=1/3) exhibits a capacity of 1 50 mAh/g when cycled in the voltage range of 3.0 V – 4.3 V. However delamination between the core and the shell are a problem to be solved. This example shows nicely, how versatile Li-NMC can be. To counteract the effect of thermal destabilization of the material of high Ni content Ni-rich NMCs can be prepared within a Mn-rich phase. Each effect can be tailored individually by the composition of the material. Nickel determines mainly the capacity, manganese the thermal stability and cobalt the rate capabilities of the material. The transition metals have different effects on the battery. For the charging process, these reactions are reversed. The oxide in the cathode is reduced during the discharging process and in the anode, graphite is oxidized. The discharging process follows the following reactions:Ĭathode: Ni xMn y Co zO 2 + Li + + e - → Li Ni xMn y Co zO 2 įigure 1: Schematic of the discharging process of a Li-ion battery. Electrons are released from the positive electrode and move externally to the anode, while the Li + ions move towards the negative electrode, forming a Li xC 6 compound at the electrode. For recharging the battery the electrodes are connected via an external electrical supply (power socket). The lithium ions released from the anode during this process move through the electrolyte and the separator to the cathode (positive electrode). During the discharging process, electrons move from the anode (negative electrode), through an external load, e.g. The discharging process is described in Figure 1. The electrolyte is needed as a contractor of the Li-ions. The electrodes have a potential difference and are kept from contact through the separator. LIBs consist of four main components, the cathode, anode, electrolyte, and separator. Li-NMC batteries are part of Li-Ion batteries (LIBs). They play an important role in a greener future through the reduction of greenhouse gases emitted by combustion cars. They are mainly used for transportation applications (e.g. Li-NMC batteries are one of the most important among LIBs and are part of the next generation. It was developed in the period of 2001-2008 in efforts to reduce the cobalt content in cathodes for LIBs while increasing the capacity of batteries. Li-NMC is a complex layered metal oxide consisting of lithium, nickel, manganese, and cobalt and has the general formula of LiNi xMn 圜o zO 2. They have a similar setup as other Li-Ion-based batteries. Li-NMC batteries have Li-NMC as cathode material and belong to the category of lithium-ion batteries (LIB). Lithium nickel manganese cobalt oxides, or short, Li-NMC, are electrode materials commonly used in rechargeable batteries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
